
About the CTU
The Copenhagen Trial Unit (CTU) is an organizational entity within the Capital Region of Denmark. It is physically located at and administratively managed by Rigshospitalet in Copenhagen, Denmark.
Explore the History and Origins of the CTU
Explore the long list of Publications from the CTU
About the CTU
The Copenhagen Trial Unit (CTU) is an organizational entity within the Capital Region of Denmark. It is physically located at and administratively managed by Rigshospitalet in Copenhagen, Denmark.
The randomised clinical trial (RCT) is the cornerstone in evaluating the beneficial and adverse effects of interventions (see The James Lind Library and Testing Treatments). Systematic reviews with meta-analyses of RCTs are acknowledged as essential tools in the evaluation of already implemented interventions, in considering new interventions to be implemented in clinical practice, or in the planning of further RCTs.
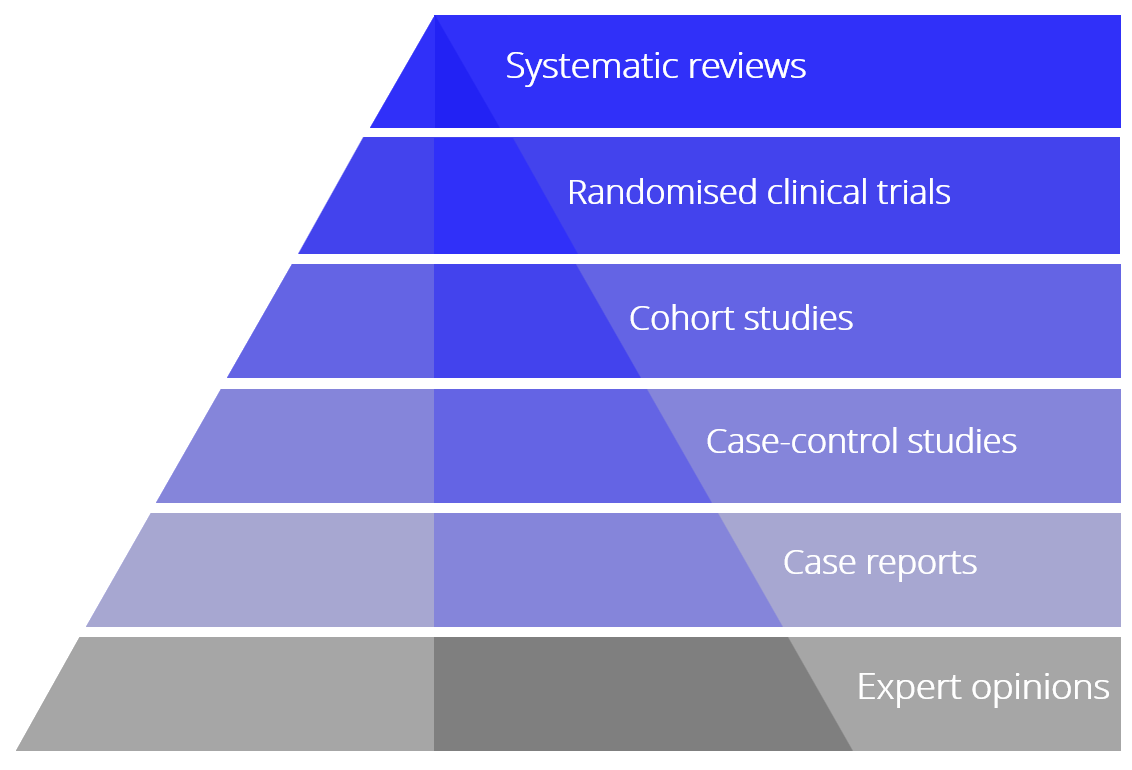
Quality of evidence
Who We Are
The Copenhagen Trial Unit (CTU) is a non-specialty oriented clinical intervention research unit. We offer flexible collaboration at all stages of clinical research as well as education in clinical trials. Trial methodology development is an indelible part of our services as it is recognised as an important tool for the quality of the clinical trial.
The core staff of the CTU consists of experienced trialists, clinicians, epidemiologists, statisticians, information-technology engineers, and information specialists.
The CTU is collaborating with the international Cochrane Collaboration in preparing, maintaining, and disseminating systematic reviews of the effects of health care. The Editorial Team Office of the Cochrane Hepato-Biliary Group (CHBG), part of the Cochrane Collaboration, is hosted within the CTU.
Furthermore, the CTU collaborates with academy and industry to constantly improve the methods of the RCT, herewith decreasing the risks of systematic errors (bias) and the risks of random errors (play of chance). In collaboration with universities and other research institutes, the CTU is running courses dealing with the various aspects of RCTs methodology and design, systematic reviews, meta-analyses, and trial sequential analyses.
Services and Collaborations
The Copenhagen Trial Unit (CTU) provides two forms of collaboration with external research group: the CTU Advisory Collaboration and the CTU Partner Collaboration
CTU Advisory Collaboration
The CTU Advisory Collaboration is available to all established research groups across all specialties in Denmark and is offered free of charge. Any research group may contact the CTU and will be offered one to three preliminary meetings to receive general guidance on the conduct of randomized clinical trials. We review the study protocol and provide advisory feedback, which the research group may use at its own discretion.

CTU Partner Collaboration
The CTU Partner Collaboration involves a longer-term collaboration in which the CTU becomes an integrated partner within the research group, irrespective of medical specialty. In such cases, the CTU is typically represented on the relevant steering committee; however, the extent and depth of its involvement may vary depending on the specific collaboration and project requirements. The funding allocated by the Danish state is not sufficient to support this level of involvement in clinical trials.
Consequently, this model generally requires that the CTU receives external funding commensurate with the specific services provided, such as methodological expertise, organizational support, data system development, data management, and statistical analysis. The CTU operates as a non-profit organization; accordingly, any required funding will be determined in accordance with this status. The CTU will consistently participate in preparing and submitting funding applications for the collaboration.

-
The CTU services include:
collaboration in the planning of randomised clinical trials;
development of trial protocols according to the SPIRIT Statement;
interactions with competent authorities and ethics committees;
randomisation solutions (web-based, staff-based telephone randomisation, etc);
data management solutions;
development of case record forms (CRFs);
coordination and project management of trials;
development of statistical analysis plan and conduct of statistical analyses;
safety reporting;
trial reporting according to the CONSORT Statement.
systematic reviewing according to the guidelines of The Cochrane Collaboration and the PRISMA Statement;
search strategies for systematic reviews;
meta-analysis and Trial Sequential Analysis (TSA);
History of the CTU
In October 1994, the Copenhagen City Council decided to establish the Copenhagen Trial Unit – the CTU. On June 1, 1995, Christian Gluud, was appointed Head of the Department.
On the 30th of April, 2025, head of department, professor Christian Gluud stepped down and on the 1st of May, 2025, chief physician, professor Janus C Jakobsen overtook this position as head of department.

Financial Structure
The CTU is funded by the Danish state which covers the basic expenses of the Unit.
These are:
investments in IT;
salaries of the core staff;
running costs;
office equipment.
The additional staff working at the CTU is paid from grants and funding related to the individual projects.
Click here to see Annual Reports

